
In the world of modern business and industry, maintaining quality and adhering to standards is paramount. A Quality Management System (QMS) serves as the framework that ensures products and services meet or exceed customer expectations and regulatory requirements. QMS encompasses various processes, procedures, and documentation that guide an organization’s quality practices. At the heart of a robust QMS lies effective technical writing, which plays a pivotal role in conveying critical information, processes, and guidelines to various stakeholders.
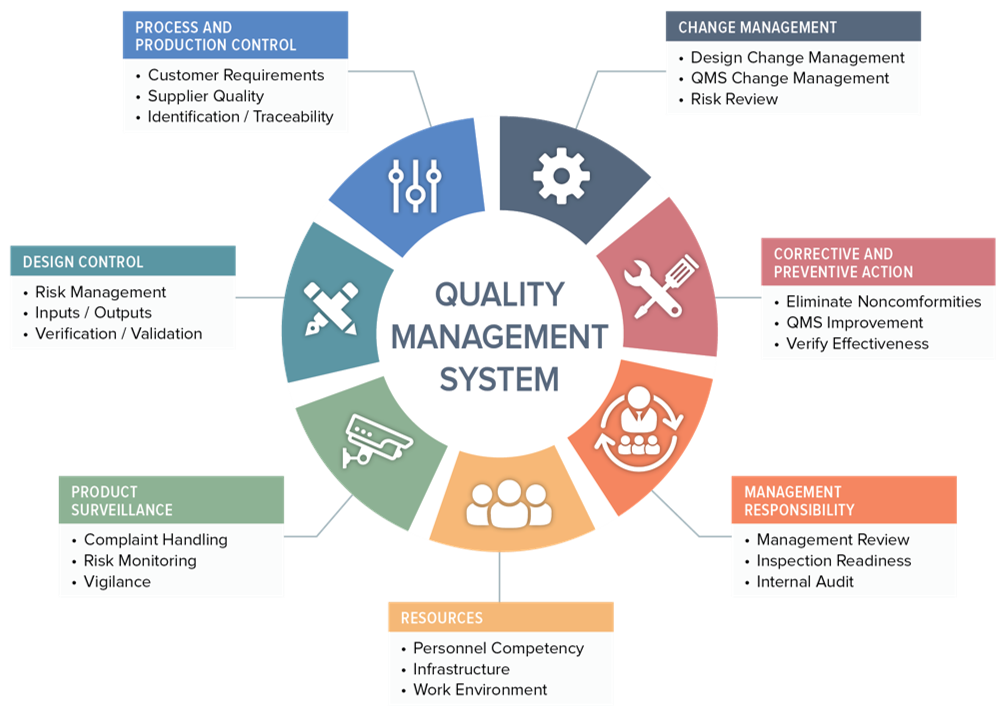
What is a Quality Management System (QMS)?
Image Source
A Quality Management System (QMS) is a structured framework that defines an organization’s policies, processes, and procedures to achieve and maintain the desired level of quality in its products or services. It involves documenting various aspects of quality control, risk management, continuous improvement, and compliance with industry standards and regulations. A well-implemented QMS helps streamline operations, enhance customer satisfaction, and ensure consistent quality output.
Also Read: Things to consider while setting up your Quality Management System
Why is Technical Writing Important for QMS?
Technical writing is the art of translating complex technical information into clear and understandable content. In the context of QMS, technical writing is crucial for several reasons:
- Clarity and Consistency: Technical documents provide precise instructions and guidelines, ensuring that processes are executed consistently across the organization.
- Knowledge Transfer: Effective documentation ensures that knowledge isn’t lost when employees leave or transition within the organization.
- Compliance: Clear documentation is essential for demonstrating adherence to regulatory standards and industry best practices.
- Risk Mitigation: Accurate documentation reduces the risk of errors, accidents, and inconsistencies that can arise due to misinterpretation.
The Role of Technical Writers in QMS
Creating and Maintaining Documentation: Technical writers play a pivotal role in creating, updating, and maintaining QMS documentation. They translate complex technical processes into comprehensive guides, manuals, and standard operating procedures (SOPs) that can be easily understood and followed by employees at all levels. Well-structured documentation minimizes errors and discrepancies, contributing to a more efficient and effective QMS.
Communicating with Stakeholders: Technical writers bridge the communication gap between subject matter experts, management, and employees. They collaborate with various stakeholders to ensure that documentation accurately represents processes, aligns with organizational goals, and meets the needs of end-users. This facilitates better understanding and implementation of quality practices throughout the organization.
Ensuring Compliance with Regulations: In regulated industries, adherence to standards and regulations is non-negotiable. Technical writers ensure that QMS documentation complies with relevant industry standards, legal requirements, and guidelines. This documentation is often subject to audits and inspections, making accuracy and compliance critical.
The Challenges of Technical Writing for QMS
Accuracy and Clarity Creating accurate and clear documentation can be challenging, especially when dealing with complex technical concepts. Technical writers must strike a balance between simplifying information for easy comprehension and retaining the necessary level of detail for accuracy.
Compliance with Regulations Industries such as healthcare, aerospace, and pharmaceuticals are subject to strict regulatory frameworks. Technical writers must stay current with evolving regulations and ensure that QMS documentation remains compliant, which can be a daunting task.
Keeping Up with Changes QMS documentation isn’t static; it evolves with changes in technology, processes, and regulations. Technical writers must continuously update documentation to reflect these changes, ensuring that employees have access to the most up-to-date information.
Best Practices for Technical Writing for QMS
Use a Template Developing documentation templates ensures consistency in format and structure. Templates provide a standardized framework for creating various types of documentation, making it easier for users to navigate and understand the content.
Get Input from Stakeholders Collaboration with subject matter experts and stakeholders helps ensure that documentation accurately represents processes and practices. This approach enhances the quality and reliability of the information presented.
Review and Revise Documentation Regularly QMS documentation isn’t a one-time endeavor. Regular reviews and revisions are essential to keep documentation accurate and up-to-date. This practice prevents the propagation of errors and outdated information.
Also Read – Quality Management System: Improve Your Organization’s Performance
Final Words
In the realm of Quality Management Systems, effective technical writing is indispensable. It acts as the connective tissue that binds complex technical processes to clear and comprehensible documentation. Technical writers play a pivotal role in creating, maintaining, and communicating QMS documentation. Their efforts not only ensure operational efficiency, consistency, and compliance but also contribute to the overall success and reputation of the organization.
At GxP Cellators, we help our clients design a robust QMS for the non-clinical and earlier clinical to later clinical stages. Our technical writers are highly skilled in designing the documentation for GCP, GLP, and GMP operations as per International Standards Organization (9001, 17025, 13485), Health Canada, U.S. Food and Drug Administration and European Medicines Agency.
We are expertized in designing the documentation for site qualification & commissioning, instrumentation qualifications, process validations, cleaning & method validations, computer system validations, vendor management & qualifications, regulatory dossiers, stability program & protocols designing, stability data request forms, and audit response to the regulatory observations.
Elevate Your Cleanroom Performance: Explore Our cleanroom commissioning and qualification services
Reference Sources:
- International Organization for Standardization (ISO): ISO 9001 Quality management
- Society for Technical Communication (STC): Importance of Technical Communication
- American Society for Quality (ASQ): Quality Management Systems
- Microsoft Writing Style Guide: Technical Writing Guidelines
- “Technical Writing for Dummies” by Sheryl Lindsell-Roberts
- “The Complete Idiot’s Guide to Technical Writing” by Krista Van Laan and Catherine Julian




4 comments
admin
16/10/2015 at 7:34 am
Lorem ipsum dolor sit amet, consectetuer adipiscing elit, sed diam nonummy nibh euismod tincidunt ut laoreet dolore magna aliquam erat volutpat.
admin
16/10/2015 at 10:26 am
Diam nonummy nibh euismod tincidunt ut laoreet dolore magna aliquam erat volutpat.
admin
16/10/2015 at 7:34 am
Lorem ipsum dolor sit amet, consectetuer adipiscing elit, sed diam nonummy nibh euismod tincidunt ut laoreet dolore magna aliquam erat volutpat.
admin
16/10/2015 at 10:26 am
Diam nonummy nibh euismod tincidunt ut laoreet dolore magna aliquam erat volutpat.