Biologics are complex products that are made from living organisms. They are often used to treat serious diseases, such as cancer and autoimmune disorders. The manufacturing process for biologics is complex and requires a high level of quality control. Biologics site qualification is a process that ensures that a manufacturing site has the necessary facilities, equipment, and procedures in place to produce biologics products that meet the required quality standards.
Let’s delve into the crucial aspects of biologics site qualification, its significance, and how it contributes to maintaining high standards in the production of life-saving medications.
Understanding Biologics Site Qualification
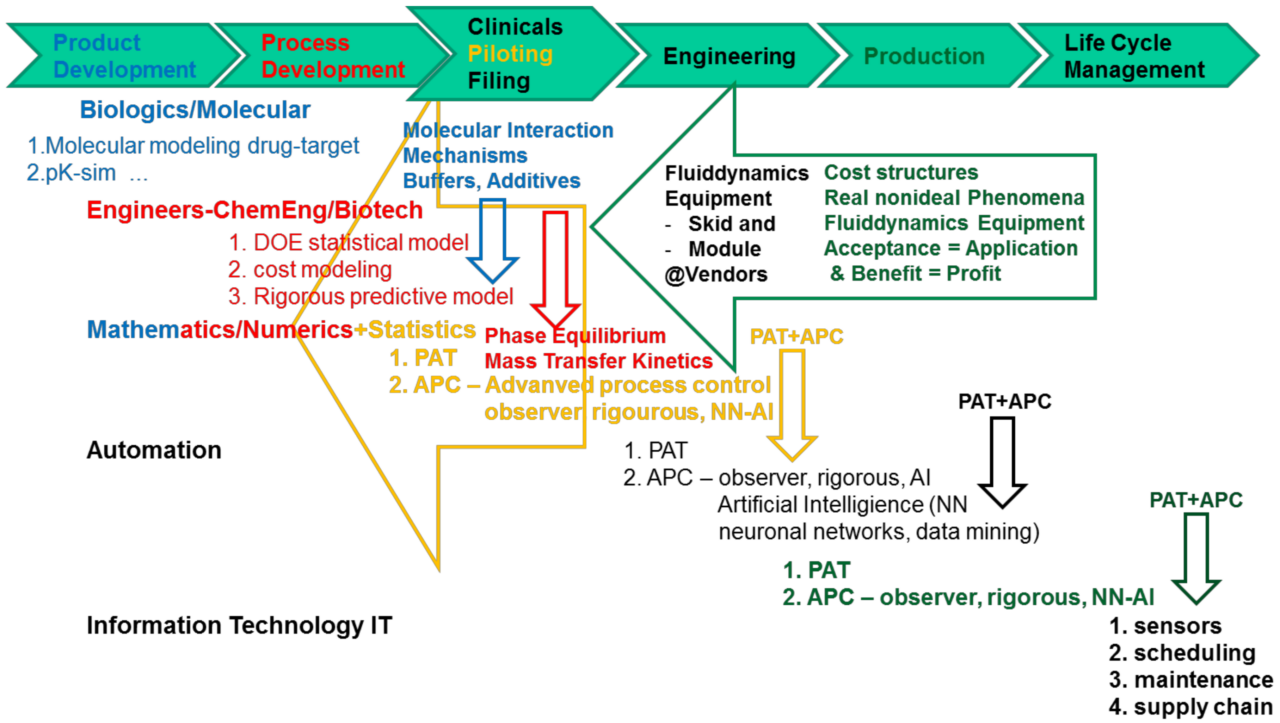
Biologics site qualification refers to the meticulous process of evaluating and certifying a manufacturing facility for the production of biopharmaceuticals. These biopharmaceuticals, also known as biologics, are complex medical products derived from living organisms. They include vaccines, blood products, gene therapies, and monoclonal antibodies. The qualification process involves rigorous assessments to ensure that the facility meets all necessary requirements to produce these intricate products.
The Significance of Biologics Site Qualification
Product Safety and Quality Assurance: Biologics are developed to treat severe diseases, and any compromise in their quality can have dire consequences. Qualifying a site guarantees that the manufacturing processes adhere to stringent quality standards, minimizing the risk of product contamination or variation.
Regulatory Compliance: Regulatory bodies, such as the Food and Drug Administration (FDA) in the United States, have stringent guidelines for the production of biopharmaceuticals. Site qualification ensures that a facility is compliant with these regulations, leading to smooth approvals for the products developed at the site.
Consistency in Production: Biologics site qualification involves assessing the consistency of manufacturing processes. Consistency is vital to ensure that every batch of biopharmaceuticals is of the same high quality, providing predictable treatment outcomes for patients.
You may like to read –
Quality Regulatory Compliance: A Guide for Businesses
Risk Management in Quality Compliance | Everything To Know
Regulatory Landscape Compliance Trends: Implications for Businesses
Key Steps in Biologics Site Qualification
Image Source : mdpi
Pre-Qualification Assessment: Before a site is qualified, a thorough assessment is conducted. This includes evaluating the facility’s infrastructure, equipment, personnel qualifications, and quality systems. Any deficiencies are identified and rectified during this stage.
Process Validation: The manufacturing processes employed at the facility undergo validation to ensure they consistently produce products that meet predetermined specifications. This involves monitoring and analyzing various parameters throughout production.
Documentation Review: A comprehensive review of the facility’s documentation, including standard operating procedures, training records, and quality control measures, is performed to ensure accuracy and adherence to regulations.
Risk Management: Potential risks that could impact product quality or patient safety are identified and mitigated. This proactive approach enhances the reliability of the facility’s operations.
Transitioning Toward Excellence
In the dynamic landscape of biopharmaceuticals, continuous improvement is essential. Biologics site qualification sets the foundation for excellence by establishing a robust framework for quality assurance and regulatory compliance. As the industry advances, the significance of maintaining high standards at every step of production becomes even more pronounced.
In conclusion, biologics site qualification is a critical process that safeguards the quality, safety, and consistency of biopharmaceutical production. It ensures that manufacturing facilities meet stringent regulatory requirements and adhere to best practices, ultimately contributing to the well-being of patients worldwide. As the biopharmaceutical field evolves, the importance of biologics site qualification
Resource: