What is cmc in pharma?
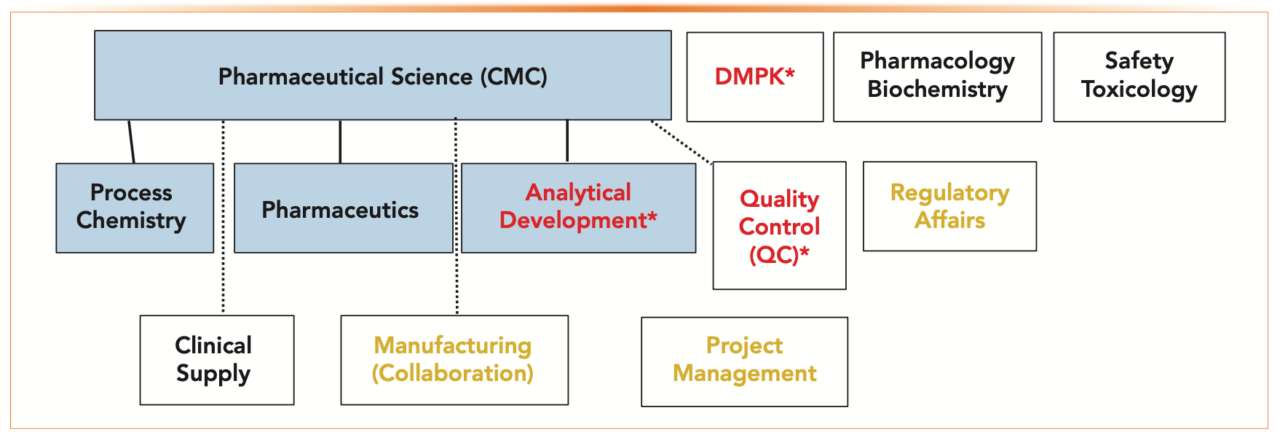
CMC stands for Chemistry, Manufacturing, and Controls. It is a critical component of drug development that ensures the quality and consistency of pharmaceutical products throughout their lifecycle. CMC activities begin early in the drug discovery process and continue through clinical trials and commercialization.
Explore Our CMC Regulatory Compliance Services
The main components of CMC are:
- Chemistry: This involves the identification, characterization, and synthesis of the drug substance. It also includes the development of analytical methods to test the quality of the drug substance.
- Manufacturing: This involves the development of processes for manufacturing the drug product, including the selection of raw materials, equipment, and manufacturing procedures. It also includes the development of controls to ensure the quality of the drug product during manufacturing.
- Controls: This involves the development of specifications and procedures to ensure the quality of the drug product throughout its lifecycle. This includes specifications for the drug substance, drug product, and packaging materials. It also includes procedures for testing, monitoring, and controlling the manufacturing process.
The following are some of the key benefits of a strong CMC program:
- Improved product quality and consistency
- Reduced risk of manufacturing defects
- Increased chances of regulatory approval
- Reduced time and cost of drug development
- Increased patient safety
GMP stands for Good Manufacturing Practices. It is a set of regulations that govern the manufacturing of pharmaceutical products. GMPs are designed to ensure the quality, safety, and efficacy of pharmaceutical products.
The main difference between CMC and GMP is that CMC is product-specific, while GMP is a set of general principles that apply to all pharmaceutical manufacturing. CMC focuses on the specific chemistry, manufacturing, and controls of a particular drug product, while GMP applies to the overall manufacturing process for all pharmaceutical products.
Also Read: GLP vs. GMP vs. GCP: Understanding the Differences
A strong CMC program is essential for the development and commercialization of safe and effective pharmaceutical products. By following GMPs, pharmaceutical companies can ensure that their products are manufactured in a consistent and controlled manner, which helps to protect patient safety.
Understanding the Significance of CMC
The significance of CMC cannot be overstated. It’s the bridge that connects scientific innovation to real-world impact. Without proper CMC practices, even the most groundbreaking discoveries might never reach the patients who need them.
The CMC Process Demystified
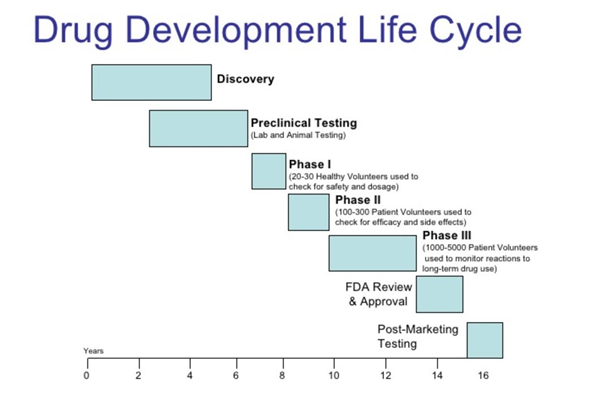
- Preclinical Phase – In the preclinical phase, researchers work on understanding the drug’s biological activity and initial safety profile. CMC considerations begin here, as the formulation’s compatibility and stability come under scrutiny.
- Clinical Development – As the drug advances to clinical trials, CMC plays a role in ensuring that the manufactured product matches the one used in trials. Consistency is key, and any changes must be carefully evaluated and documented.
- Regulatory Submission – Regulatory bodies require comprehensive CMC data as part of the drug approval process. This data substantiates the drug’s quality, safety, and consistency.
- Commercial Production – When the drug receives approval, scaling up production becomes a challenge. CMC practices ensure that larger batches maintain the same quality as those used in trials.
- Ensuring Quality through CMC – CMC serves as a safeguard against variability. It ensures that each dose of medication, whether produced today or years from now, is of the same high quality.
- Regulatory Authorities and CMC Guidelines – Regulatory agencies like the FDA provide guidelines for CMC practices. These guidelines harmonize the industry’s approach to CMC, promoting global consistency.
- Navigating Challenges in CMC Implementation – Implementing CMC isn’t without hurdles. Balancing innovation with consistency, managing supply chain complexities, and staying compliant with evolving regulations are some challenges.
Innovations in CMC: Embracing Technological Advances
The pharmaceutical industry is constantly evolving, and technological advances are playing a major role in this transformation. In the area of CMC, there are a number of innovative technologies that are being used to improve the quality, safety, and efficiency of drug development and manufacturing.
Some of the most promising technological advances in CMC include:
- Artificial intelligence (AI) and machine learning (ML): AI and ML are being used to automate tasks in CMC, such as data analysis, process optimization, and quality control. This can help to improve efficiency and reduce costs.
- Big data analytics: Big data analytics is being used to collect and analyze large amounts of data from CMC processes. This data can be used to identify trends, improve decision-making, and prevent problems.
- 3D printing: 3D printing is being used to create prototypes of drug products and manufacturing equipment. This can help to speed up the development process and reduce costs.
- Virtual reality (VR) and augmented reality (AR): VR and AR are being used to train CMC staff and simulate manufacturing processes. This can help to improve safety and efficiency.
- Cybersecurity: Cybersecurity is a critical issue in CMC, as pharmaceutical companies need to protect their data from cyberattacks. New technologies are being developed to improve cybersecurity in CMC.
These are just a few of the technological advances that are being used in CMC. As these technologies continue to develop, they will have a major impact on the way that drugs are developed and manufactured.
Also Read: Future of CMC Consultancy: Emerging Trends and Technologies
Here are some specific examples of how these technologies are being used in CMC:
- AI is being used by Novartis to develop a new drug for Alzheimer’s disease. The company is using AI to analyze data from clinical trials to identify patterns that could help to improve the drug’s efficacy.
- Pfizer is using ML to optimize the manufacturing process for its COVID-19 vaccine. The company is using ML to identify ways to improve the efficiency of the manufacturing process and reduce costs.
- Johnson & Johnson is using big data analytics to track the safety of its vaccines. The company is using big data analytics to collect data from clinical trials and post-market surveillance to identify any potential safety concerns.
- Genentech is using 3D printing to create prototypes of new drug products. The company is using 3D printing to speed up the development process and reduce costs.
- Sanofi is using VR to train its staff on how to manufacture vaccines. The company is using VR to create realistic simulations of the manufacturing process, which helps to improve safety and efficiency.
These are just a few examples of how technological advances are being used in CMC. As these technologies continue to develop, they will have a major impact on the way that drugs are developed and manufactured. This will lead to improved quality, safety, and efficiency, which will ultimately benefit patients.
Technological advancements, such as process automation and real-time monitoring, are revolutionizing CMC. These innovations enhance efficiency and data integrity.
CMC and Drug Life Cycle Management
CMC data is an important part of regulatory submissions for new drugs. The FDA and other regulatory agencies require pharmaceutical companies to provide CMC data to demonstrate the safety and efficacy of their products.
Drug life cycle management (DLCM) is the process of managing a drug from its development to its discontinuation. It includes activities such as:
- Regulatory affairs: This involves managing the regulatory process for a drug, from getting approval to ensuring compliance with regulations.
- Market access: This involves getting a drug to market and making it available to patients.
- Commercialization: This involves marketing and selling a drug to patients and healthcare providers.
- Patient safety: This involves monitoring the safety of a drug and managing any risks.
- Product life extension: This involves extending the life of a drug by finding new uses for it or developing new formulations.
CMC is an essential part of DLCM. By ensuring the quality and consistency of pharmaceutical products, CMC helps to protect patient safety and ensure the success of drugs throughout their lifecycle.
Here are some of the ways that CMC can be used to improve drug life cycle management:
- By developing robust and reliable manufacturing processes, CMC can help to ensure that drugs are produced consistently and meet quality standards. This can help to reduce the risk of manufacturing defects and ensure that patients receive safe and effective drugs.
- By developing effective quality control procedures, CMC can help to ensure that drugs are tested and monitored throughout their lifecycle. This can help to identify and address any potential quality issues early on, before they cause harm to patients.
- By providing comprehensive data on the chemistry, manufacturing, and controls of drugs, CMC can help to support regulatory approvals and ensure that drugs are compliant with regulations. This can help to ensure that drugs are available to patients in a timely manner and that they are used safely and effectively.
- By providing information on the risks and benefits of drugs, CMC can help to support patient decision-making and ensure that drugs are used appropriately. This can help to improve patient safety and ensure that drugs are used to their full potential.
Overall, CMC is a critical component of drug life cycle management. By ensuring the quality and consistency of pharmaceutical products, CMC helps to protect patient safety and ensure the success of drugs throughout their lifecycle.
CMC isn’t a one-time consideration. It’s a continuous process that extends throughout the drug’s life cycle, ensuring its quality and safety as manufacturing processes evolve.
Conclusion
In the intricate realm of pharmaceuticals, CMC shines as a guiding light that ensures quality, consistency, and patient safety. From the initial stages of drug development to its final manufacturing, CMC is the backbone that supports innovation and transforms scientific discoveries into tangible solutions for the world’s health challenges.
FAQ
Q1: Why is CMC important in the pharmaceutical industry?
Answer: CMC is crucial in the pharmaceutical industry because it ensures the consistent quality, safety, and efficacy of pharmaceutical products. It encompasses the chemistry of the drug substance, the manufacturing processes, and the controls in place to maintain product quality. Proper CMC documentation and adherence to guidelines help ensure that products are consistently produced to meet the required standards.
Q2: How does CMC impact the drug development process?
Answer: CMC plays a significant role in the drug development process. It outlines the manufacturing procedures, specifications, and controls that must be established to produce a pharmaceutical product. CMC information is submitted to regulatory agencies as a part of the drug approval process. If the CMC aspects are not well-documented or do not meet regulatory standards, it can lead to delays in approval or even rejection of the product.
Q3: Are there global guidelines for CMC implementation?
Answer: Yes, there are global guidelines for CMC implementation. Regulatory authorities such as the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and others have established guidelines that outline the requirements for CMC documentation, manufacturing processes, and quality control. These guidelines ensure consistency and quality in pharmaceutical products worldwide.
Q4: What role does technology play in advancing CMC practices?
Answer: Technology plays a crucial role in advancing CMC practices. Advanced analytical tools, automation, data management systems, and process modeling technologies enable pharmaceutical companies to monitor and control manufacturing processes more precisely. This results in increased efficiency, reduced variability, and better product quality. Additionally, technology aids in real-time monitoring of manufacturing conditions, helping to detect and address deviations promptly, thus enhancing overall CMC compliance.
Further Reading