
Risk Management in the world of industries and regulations, ensuring the quality and safety of products and services is of paramount importance. This is where risk management comes into play. It is a structured approach that enables organizations to identify, assess, and control potential risks that could jeopardize their operations, compliance, and reputation.
This article explores the significance of risk management in quality compliance, the types of risks that can impact compliance, the process, relevant tools and techniques, and its role in achieving compliance.
What is Risk Management?
Risk management is the systematic process of identifying, assessing, mitigating, and monitoring potential risks that could affect an organization’s objectives. It involves recognizing uncertainties that could lead to negative outcomes and taking proactive measures to minimize their impact.
Importance
Quality compliance refers to adhering to established standards and regulations to ensure that products and services meet predetermined quality criteria. Risk management is crucial in this context because:
- Preventing Non-Compliance: It helps identify potential risks that could lead to non-compliance with quality standards and regulations.
- Protecting Reputation: Maintaining compliance enhances an organization’s reputation for reliability and quality, and effective risk management safeguards this reputation.
- Avoiding Financial Losses: Non-compliance can result in fines, litigation, and product recalls, leading to substantial financial losses. It minimizes the likelihood of such events.
Types of Risks Affecting Quality Compliance
Several types of risks can impact quality compliance:
- Operational Risks: These arise from internal processes and systems that fail to meet quality standards.
- Regulatory Risks: Resulting from non-compliance with industry regulations and standards.
- Supply Chain Risks: Issues within the supply chain that can affect the quality of materials or components.
- Market Risks: Fluctuations in demand, competition, or customer expectations that impact product quality.
You May would like to read –
Quality Regulatory Compliance Guide for Businesses
Quality Lifecycle Management: Improve Quality & Compliance
Implementing GxP Regulatory Best Practices
GxP Regulatory Inspections: What You Need to Know?
GxP Compliance Software: Ensuring Quality & Regulatory in Life Sciences
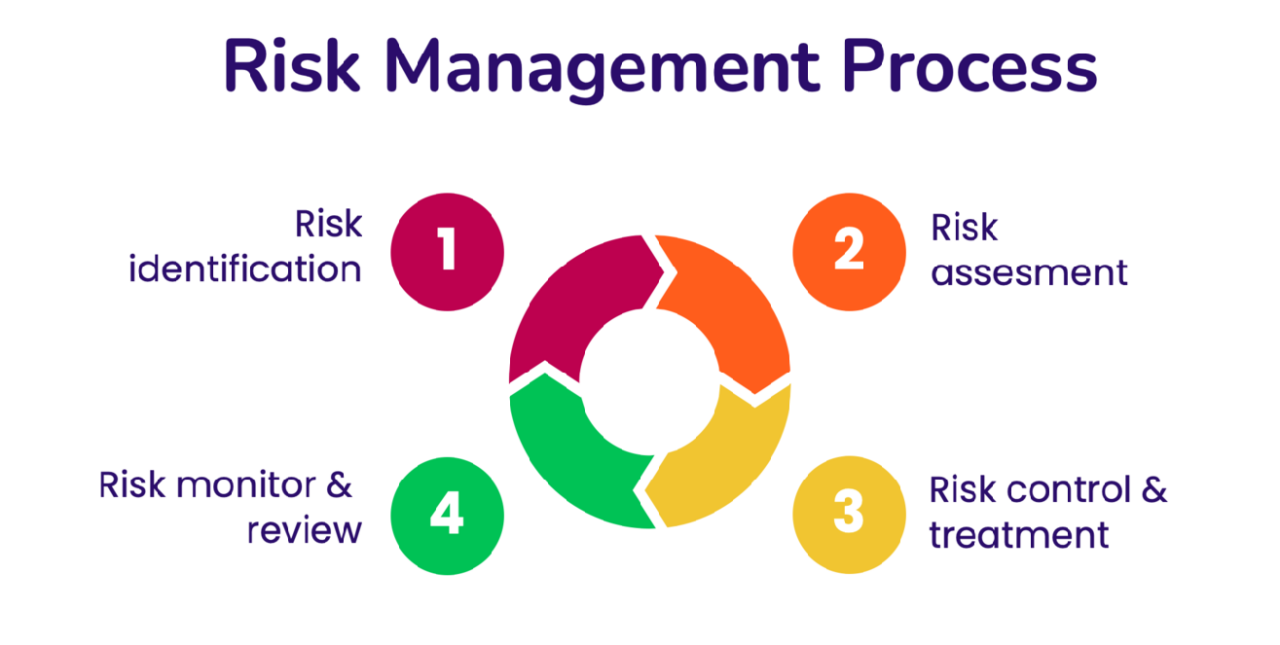
The process consists of several key steps:
- Identify Risks: Recognize potential risks that could affect quality compliance.
- Assess Risks: Evaluate the likelihood and potential impact of identified risks.
- Control Risks: Develop strategies to mitigate or eliminate identified risks.
- Monitor and Review Risks: Continuously monitor the effectiveness of risk control measures and update them as needed.
Tools and Techniques
Several tools aids:
- Hazard Analysis and Critical Control Points (HACCP): Common in the food industry, HACCP identifies and controls hazards that could compromise product safety and quality.
- Failure Mode and Effects Analysis (FMEA): A structured approach to identifying potential failure modes in processes, products, or systems and their potential impacts.
- Root Cause Analysis (RCA): Focuses on identifying the underlying causes of issues or failures to prevent recurrence.
Role of Risk Management in Quality Compliance
Image: Source
Achieving Compliance: It helps organizations align their processes with regulations, reducing the likelihood of non-compliance.
Benefits of Implementation: Implementing a robust system enhances overall operational efficiency, reduces losses, and boosts customer trust.
Conclusion
In the intricate landscape of quality compliance, risk management serves as a guiding light. By identifying, assessing, and mitigating risks, organizations can navigate the regulatory landscape more effectively, ensuring that their products and services meet the highest quality standards. In doing so, they not only protect their bottom line but also secure their reputation as purveyors of quality.
Improving Risk Management in Quality Compliance
To enhance quality compliance, organizations should:
- Foster a culture of risk awareness and proactive problem-solving.
- Invest in training to equip employees with risk management skills.
- Regularly update risk assessments to adapt to changing circumstances.
- Utilize advanced technologies to streamline risk identification and management processes.
For those seeking to delve deeper into quality compliance, the following resources are invaluable:



2 comments
online casino complaints
18/07/2024 at 3:46 am
Wow! Finally I got a website from where I be able to truly
obtain helpful information regarding my study and knowledge.
Review my web page; online casino complaints
Pingback: SOP Writers Documentation | QMS | SOP Writers